Hydrogen production through electrochemical water splitting powered by renewable electricity is a critical process for building a sustainable hydrogen economy.
Advancing alkaline water electrolysis (ALKWE) to achieve ampere-level current densities with reduced energy consumption is a central objective. However, this goal is hindered by the trade-off between activity and stability in the hydrogen evolution reaction (HER), which is exacerbated by the violent evolution of hydrogen bubble at high current densities. These bubbles disturb mass transport, block active sites, and lead to catalyst layer peeling.
An atomic-to-macroscale assembled Ni/MoO2 electrode for alkaline water electrolysis (Image by JIANG Shang)
In a study published in the Journal of the American Chemical Society, a team led by Prof. DENG Dehui, Associate Prof. LIU Yanting, and Prof. YU Liang from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) proposed an atomic-to-macro multiscale electrode design strategy. By constructing monolithic electrodes with hierarchical porous structures, the team achieved high-efficiency and long-life hydrogen production at ampere-level current densities.
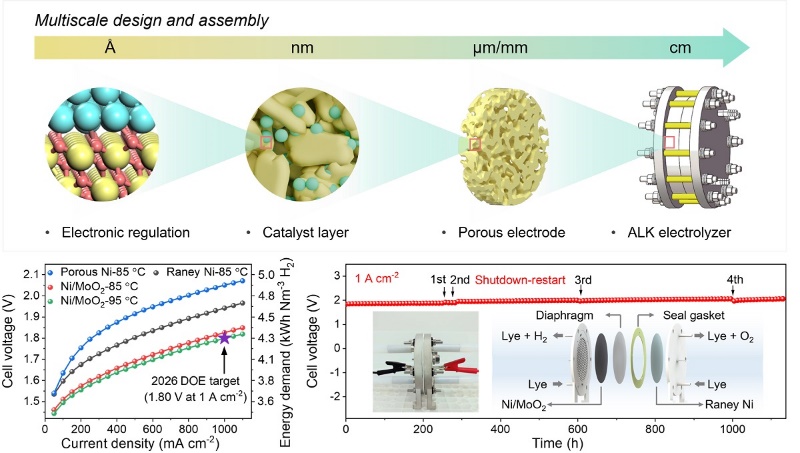
The team constructed a monolithic electrode (Ni/MoO2) featuring abundant atomic heterointerfaces and tri-scale (nano-micro-macro) porosity. This was achieved through the in situ growth of Ni nanoparticle-anchored MoO2 onto a porous Ni framework prepared via powder metallurgy.
The team found that the electrode exhibits a triple-enhancement effect in water electrolysis. First, interfacial electron transfer from Ni to MoO2 moderately weakens H* adsorption and promotes H2 desorption, thereby enhancing intrinsic activity. Second, the tri-scale hierarchical porosity, together with the hydrophilic MoO2 coating, accelerates bubble detachment and promotes electrolyte permeation, improving mass transfer. Finally, strong interactions between Ni and MoO2, along with their robust integration into the electrode skeleton, enhance structural stability.
As a result, the electrode achieves an overpotential of 145 mV at 1 A cm-2 in 1 M KOH—lower than the 300 mV required for commercial Pt/C catalysts—while maintaining stable operation for more than 3,500 hours. In practical alkaline electrolyzer tests under industrial conditions (30 wt% KOH at ≥ 85 °C), the electrode achieves a cell voltage of 1.80 V with an energy consumption of 4.3 kWh Nm-3 H2 at 1 A cm-2, with operational durability exceeding 1,000 hours.
"This atomic-to-macro multiscale electrode design strategy overcomes the long-standing dilemma in high-current-density ALKWE caused by the activity-stability tradeoff, providing an important advancement for sustainable hydrogen production," said Prof. DENG.