Ethylene oxide (EO) is a highly versatile chemical intermediate widely used in the production of polymers, solvents, and other industrial chemicals. Currently, industrial EO production relies on the epoxidation of ethylene with oxygen over silver-based catalysts., conducted at temperatures of 230–270 °C and pressures of 1–3 MPa. However, this process is plagued by limited ethylene conversion and EO selectivity, along with substantial energy consumption and CO2 emission.
Electrocatalytic ethylene epoxidation driven by renewable electricity and using water as the sole oxidant, offers a green and low-carbon alternative. Yet, the efficiency of this reaction is constrained by a fundamental trade-off between ethylene conversion and EO selectivity.
To address this challenge, a research team led by Profs. BAO Xinhe and GAO Dunfeng from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), in collaboration with Prof. WANG Guoxiong from Fudan University, has developed a novel strategy for direct electrocatalytic ethylene epoxidation. By stabilizing superoxo species with platinum (Pt) single atoms, the team enhanced EO production efficiency.
Their findings were recently published inJournal of the American Chemical Society,
Single atom Pt-stabilized superoxo species for direct electrocatalytic ethylene epoxidation (Image by WANG Hanyu)
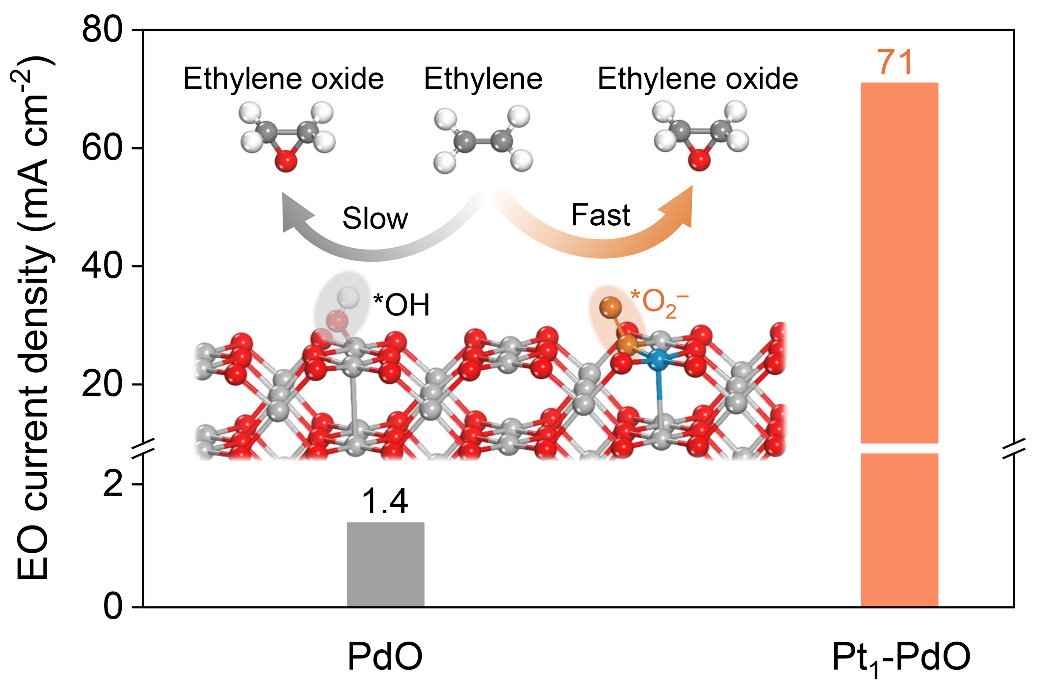
The team designed a catalyst consisting of Pt single atoms supported on palladium oxide (PdO), which facilitates the formation of superoxo species as reactive oxygen species (ROS) and accelerates the selective oxidation of ethylene. This catalyst enables efficient EO production in a zero-gap anion exchange membrane-based membrane electrode assembly (MEA) electrolyzer, achieving a Faradaic efficiency of 74% and a partial current density of 71 mA cm–2.
In addition, the researchers conducted a scale-up demonstration using a 100cm2 electrolyzer, attaining an EO production rate of 6.4 g h–1 at a total current of 15 A. This result highlights the practical application potential of this catalyst design for electrochemical ethylene valorization.
Furthermore, the study revealed that Pt single atoms, in synergy with perchlorate anion in the electrolyte, promote the formation of abundant superoxo species and accelerate the epoxidation of ethylene activated on the PdO support.
"Our study underscores the importance of generating and stabilizing specific ROS for the selective oxidation of organic molecules," said Prof. GAO. "And paving the way for more efficient and sustainable chemical synthesis processes".