Formate is a key chemical building block widely used in industries. Converting carbon dioxide (CO2) into formate through hydrogenation offers a promising route to transform greenhouse gas into high-value chemicals. However, while heterogeneous catalysts based on non-precious metals have been explored for this reaction, their practical application has been limited by low intrinsic reactivity.
Confining Co atoms into the MoS2 lattice enables highly efficient hydrogenation of CO2 to formate (Image by WANG Zifeng and HU Jingting)
In a study published in Nature Communications, a research group led by Prof. DENG Dehui, Prof. YU Liang, and Assoc. Prof. HU Jingting from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) developed a novel catalyst that achieves high-efficiency CO2 hydrogenation to formate.
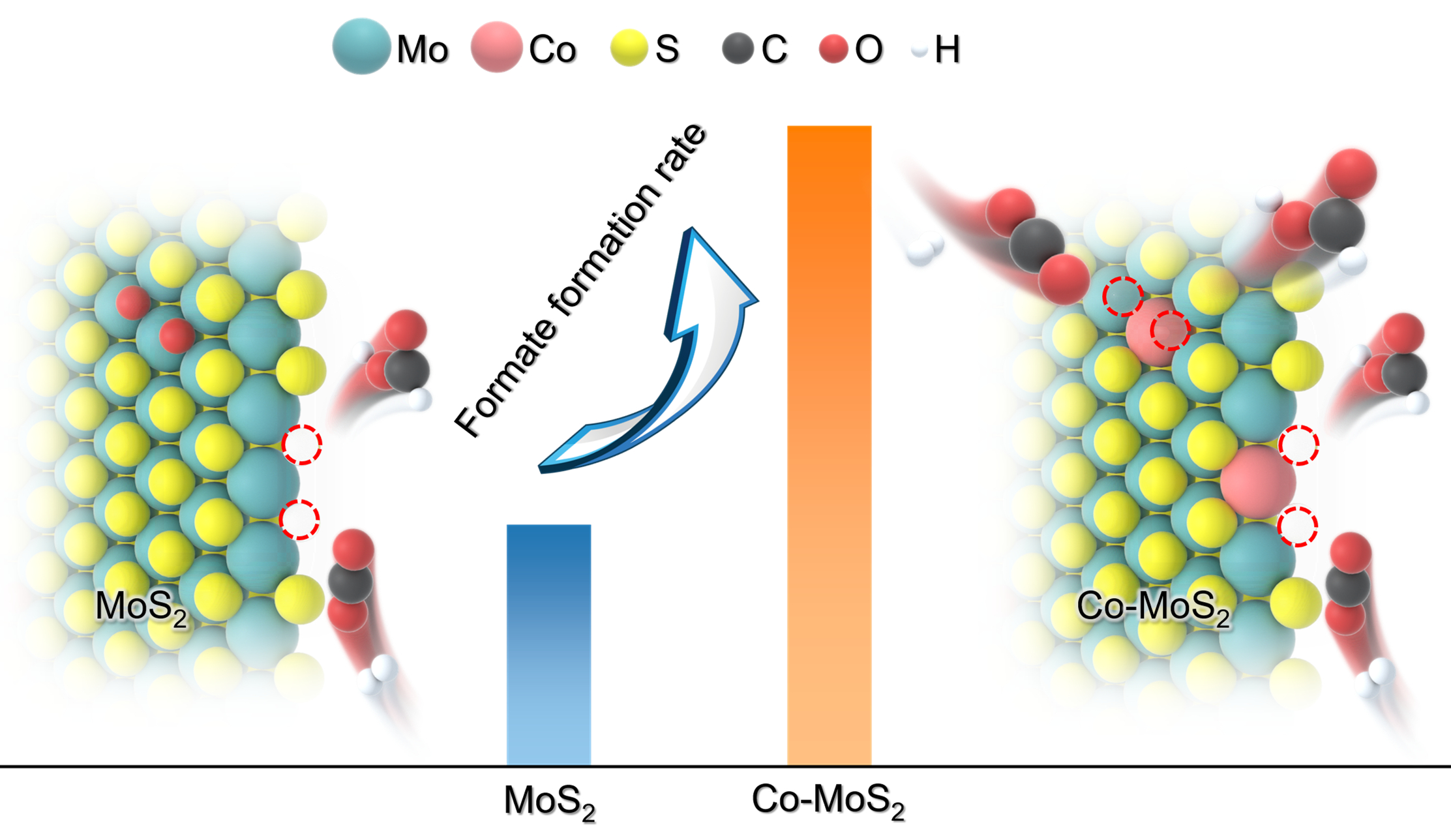
The researchers confined cobalt (Co) atoms into the MoS2 lattice to create highly active sulfur vacancy (SV)-confined Co-Mo sites. They found that the lower coordination number of Co compared to Mo weakens the bonding interaction between the lattice-confined Co-Mo pair and surface sulfur or oxygen. This facilitated the hydrogenation-driven removal of sulfur or oxygen species, generating SVs. These exposed SV-confined Co-Mo sites, with moderate CO2 adsorption at both edges and basal planes, suppressed C-O bond cleavage and favored formate formation, resulting in the superior catalytic activity and selectivity.
The Co-MoS2 catalyst achieved a formate production rate of 17.0 mmol gcat.-1 h-1 with over 99% selectivity at 200 °C—nearly three times than pristine MoS2. Moreover, its activity remained stable for more than 80 hours over eight reaction cycles, demonstrating high durability.
"This work offers a promising method for generating abundant oxygen-tolerant SVs by confining Co in the MoS2 lattice to modulate its electronic structure, improving the catalytic performance of CO2 hydrogenation to formate," said Prof. DENG.