Aluminas, particularly γ‑Al2O3, are widely used in modern energy and environmental industries, serving as both catalysts and catalyst supports. Halogen-modified alumina is often employed to achieve superior catalytic performance, as the incorporation of halogen elements can substantially alter its surface chemical properties. In particular, introducing fluorine or chlorine can modify surface acidity. However, the atomic-scale origin of this enhanced acidity has remained controversial for decades due to the complexity of halogenated alumina and the challenges associated with its spectroscopic characterizations.
Recently, a research team led by Prof. HOU Guangjin and Prof. JI Yi from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has revealed the strong Brønsted acid site (BAS) in fluorinated γ‑Al2O3 using state-of-the-art solid-state nuclear magnetic resonance (NMR) techniques.
The study was published in Journal of the American Chemical Society.
Single-site level identification of strong Brønsted acid site in fluorinated Al2O3 revealed by advanced solid-state nuclear magnetic resonance (Image by Zhang Huizhen)
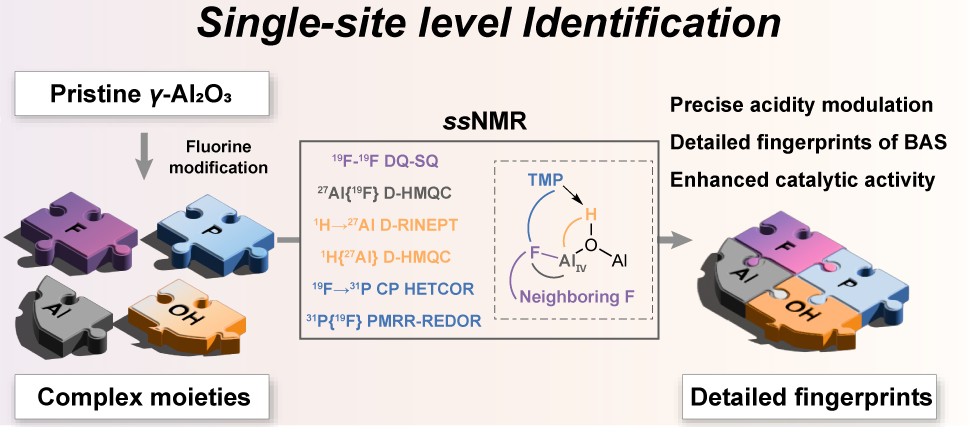
The researchers employed advanced solid-state NMR techniques, combining high magnetic fields (up to 18.8 T), ultrafast magic-angle spinning (MAS, up to 60 kHz), and multinuclear multidimensional correlation experiments (1H‑27Al, 19F‑27Al, 19F‑31P, etc.), complemented by trimethylphosphine (TMP) probe adsorption. This comprehensive approach enabled precise identification of the structural origin of strong Brønsted acid sites in fluorinated γ-Al2O3.
They found that strong Brønsted acid sites are present only on fluorinated γ‑Al₂O₃ and are absent on chlorinated analogues. These sites were unambiguously identified as bridging hydroxyl groups bound to a stable, monofluoride-incorporated tetracoordinated aluminum center, denoted as F1−AlIV−μ2−OH. Notably, the 1H, 19F, and 27Al NMR signatures of these sites converge with those of bridging acid sites in fluorinated zeolites, revealing a previously unrecognized structural similarity between the two classes of materials.
Further experiments demonstrated that these strong Brønsted acid sites exhibit exceptional robustness, remaining intact after exposure to air, moisture, and even liquid water washing. Catalytic tests using 1‑octadecene conversion demonstrated that the presence of these sites enhances catalytic activity and aromatization performance, directly linking the atomic-scale structure to macroscopic catalytic behavior.
"Our work resolves the long-standing controversy on the nature of strong acidity in halogenated aluminas and establishes a structural benchmark for the targeted design of fluorinated catalysts," said Prof. HOU.