Boron oxide is a fundamental component in a wide range of materials, from everyday glass to advanced optics and high-energy propellants. Under ambient pressure, bulk B2O3 adopts a vitreous structure characterized by a two-dimensional network. However, the molecular-level mechanisms underlying the formation and growth of this network remain unclear.
Neutral boron oxide clusters provide ideal model systems for investigating these structural evolutions. Yet, experimental studies have long been hindered by the difficulty of size selection, as neutral clusters lack charge and are challenging to isolate and characterize.
In a study published in the Journal of the American Chemical Society, a research team led by Prof. JIANG Ling and Prof. LI Gang from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), in collaboration with Prof. Lai-Sheng Wang from Brown University, has experimentally discovered that neutral boron oxide clusters (BO3, B2O4, B3O6) contain key structural units (BO, BO3, B2O5), which are essential building blocks of the two-dimensional vitreous network. The findings provide important insights into the microstructure and growth mechanisms of vitreous materials.
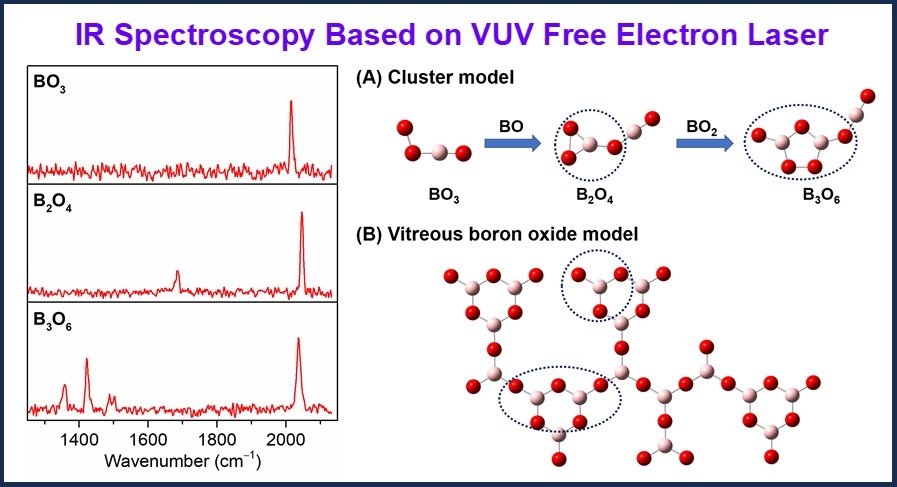
Experimental IR spectra via the tunable vacuum ultraviolet free electron laser, identified planar structures of neutral boron oxide clusters, and a vitreous boron oxide model (Image by LI Shangdong and JIANG Shuai)
Previously, the team developed a neutral-cluster infrared spectroscopy station based on the Dalian Coherent Light Source, which enabled high-sensitivity dedicated infrared (IR) spectral detection, structural characterization, and reactivity studies of mass-selected neutral clusters. By combining this platform with a tunable vacuum ultraviolet free-electron laser (VUV-FEL) at the Dalian Coherent Light Source, the team obtained size- selected IR spectra of neutral BO3, B2O4, and B3O6 clusters.
Comparison between the experimental spectra and high-level quantum chemical calculations reveals that all three clusters adopt planar structures. Structural analysis shows that BO3, B2O4, and B3O6 are composed of BO, BO3, and B2O5 units, respectively—key structural units that constitute the vitreous two-dimensional network. Further chemical bonding analyses indicate that the structural stability of these clusters arises from the synergy among the terminal B≡O groups and B–O σ bonds.
This work not only provides crucial spectral signatures for the key structural units of bulk boron oxides but also serves as a robust model system. The findings pave the way for systematic, size-dependent studies of larger systems to understand the stepwise formation and growth mechanisms of two-dimensional boron oxide networks.