Organic halides exhibit distinct biological activities and reactivity, making them indispensable in pharmaceutical development, energy materials, and functional molecule design. Their critical role in molecular innovation continues driving the developing of new synthetic methodologies.
Although direct alkene halogenation represents a classical route to organic halides, conventional methods are limited to the formation of vicinal dihalides, which limits the structural diversity and synthetic utility of these compounds.
Recently, a research team led by Prof. CHEN Qing-An from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), developed a phosphordiamidate-catalyzed strategy for the highly selective synthesis of 1,3-, 1,4- and 2,3-dihalogenation products. The study, published in Journal of the American Chemical Society, providing a new way for constructing complex organic halides.
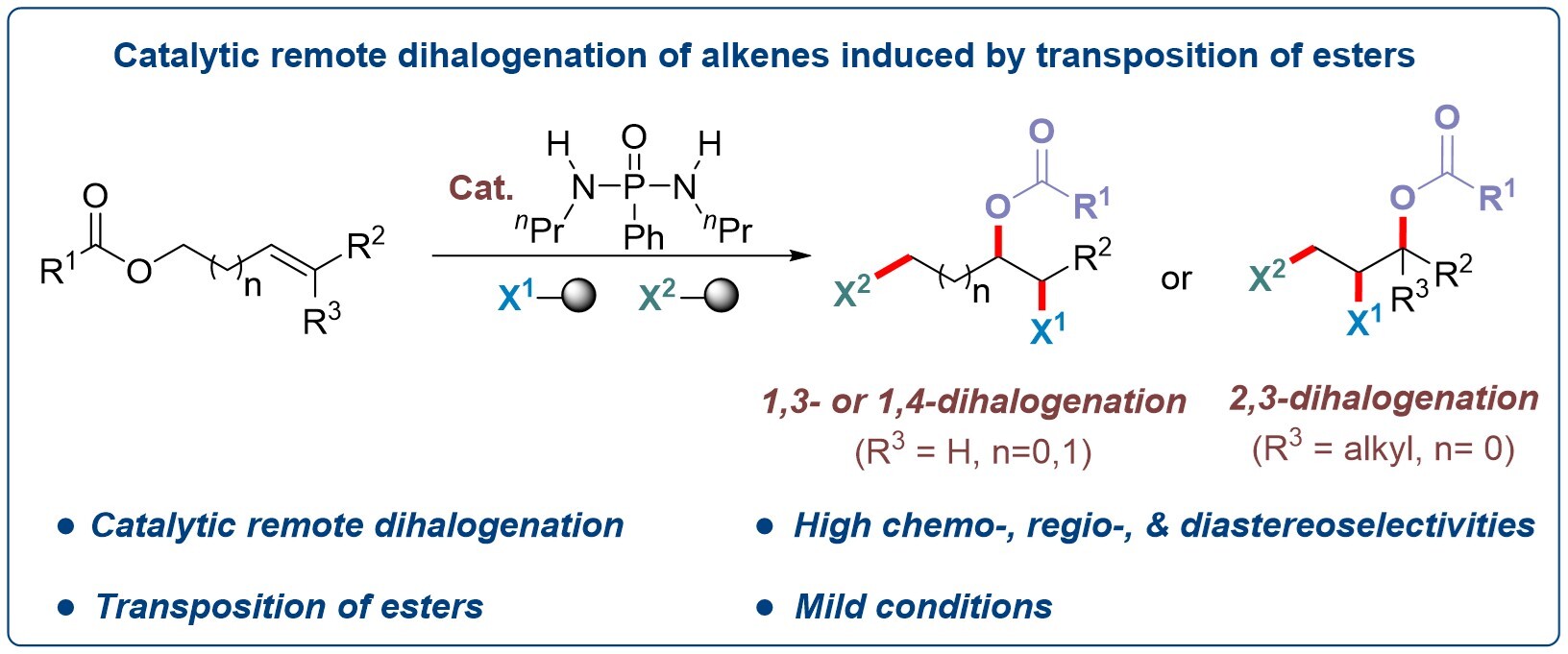
A strategy for ester-transposition-induced remote dihalogenation of alkenes (Image by Chang-Hui Liu)
The researchers developed a phosphordiamidate-catalyzed, directing-group-free strategy for remote dihalogenation of alkenes. Regioselective was achieved through an ester transposition process. Under mild conditions, the catalyst cooperates with NBS and SOCl2 to generate active intermediates, which then react with allylic or homoallylic esters to afford 1,3-, 1,4-, and 2,3-dihalides with high efficiency and selectivity.
The researchers demonstrates broad substrate compatibility including various unactivated alkenes, along with good functional group tolerance such as cyano and hydroxyl groups. Gram-scale experiments supported the practicality of the strategy. Besides, the resulting products could undergo diverse derivatizations including cross-coupling and cyclization reactions, highlighting their potential utility in complex molecule construction and pharmaceutical synthesis.
"This work may inspire the development of transposition-induced remote difunctionalizations strategies," said Prof. CHEN