Alkali and alkaline earth metal hydrides hold great promise for hydrogen storage and hydrogen-involved chemical transformations due to the unique properties of hydridic hydrogen (H‾). However, bulk hydrides often suffer from high lattice energy and limited exposure of active sites, hindering their catalytic performance.
Recently, a research group led by Prof. GUO Jianping and Prof. CHEN Ping from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), in collaboration with Prof. CHANG Fei from Yongjiang Laboratory and Prof. RAO Li from Central China Normal University, developed atomically dispersed barium hydride catalysts for the synthesis of deuterated alkylarenes. This study was published inNature Communications.
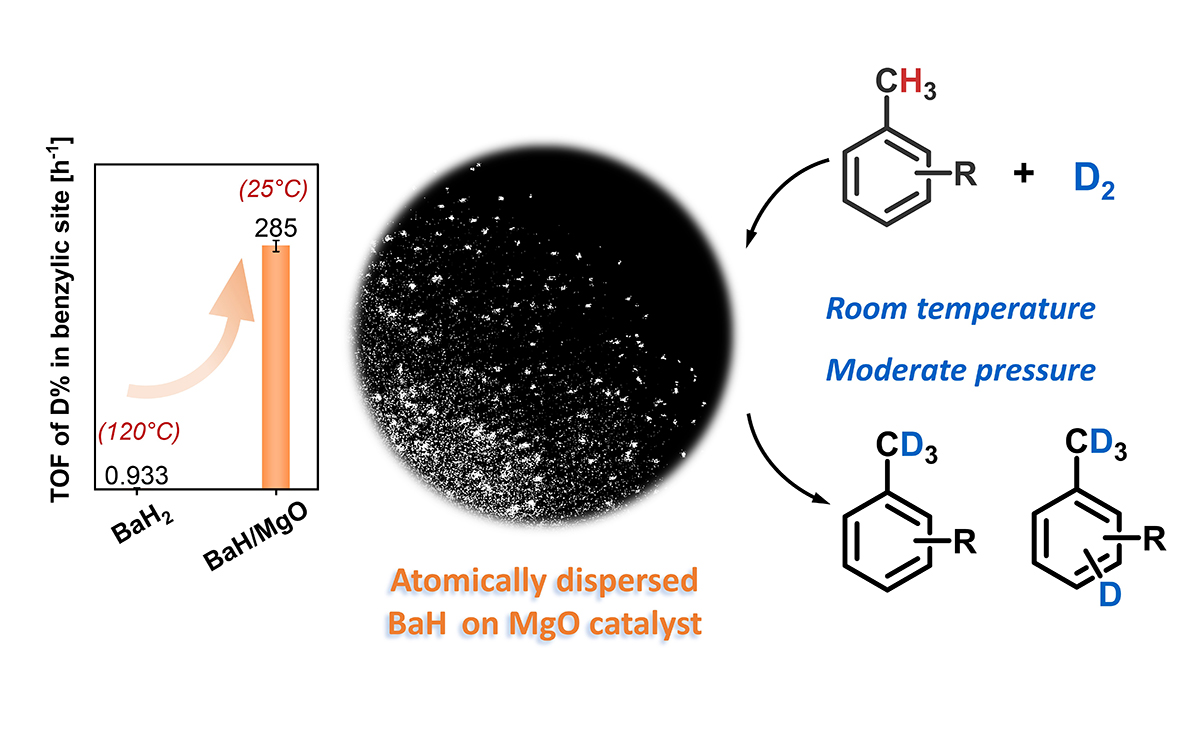
Efficient deuteration of alkylarenes catalyzed by atomically-dispersed barium hydride materials (Image by CAI Yongli)
Researchers synthesized atomically dispersed barium hydride catalysts on magnesium oxide (BaH/MgO) using a convenient impregnation-hydrogenation method. This (sub)nanostructured hydride material acts as an efficient, transition metal-free heterogeneous catalyst for hydrogen activation and hydrogen isotope exchange reactions across a range of nonactivated alkylarene substrates.
The catalyst enabled regioselective deuteration of benzylic sites in toluene and its derivatives, achieving a high turnover frequency that surpasses both homogeneous and heterogeneous counterparts. Besides, it facilitated the deuteration of non-directional aromatics at room temperature with efficiency comparable to transition metal-catalyzed processes, which typically require elevated temperatures.
Theoretical studies revealed that the surface barium hydride species play a multifaceted role, enabling deprotonation of sp3C-H bonds and nucleophilic attack on the sp2C atoms to yield regioselective products.
"This study offers an alternative synthetic route for the production of deuterium-labeled compounds and shows potential for further applications in tritium labeling of alkylarenes," said Prof. CHEN.
"This new method for preparing (sub)nanostructured metal hydrides may open new avenues for their applications in diverse chemical processes," said Prof. GUO.