Photocatalysis involves three fundamental steps: light absorption, charge separation and transfer, and chemical reactions. These reactions occur at the solid-liquid interface where the complex charged environment influences reaction kinetics. Most research has focused on the charge transfer processes within solid catalysts.
Understanding the interplay between surface charges and charge transfer at the catalyst-electrolyte interface is critical for advancing photocatalysis. However, directly measuring surface charges in electrolytes at the nanoscale has long been a challenge.
Recently, Prof. FAN Fengtao and Prof. LI Can et al. from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) have developed an innovative approach to measure surface charges in liquid environments. This study was published in the Journal of the American Chemical Society.
By using a charged probe to isolate electrostatic forces from long-range interactions, researchers successfully mapped the electric field distribution within the electrical double layer. This method enabled direct measurement of surface potential and photovoltage under realistic liquid conditions. Moreover, they uncovered a key phenomenon: Surface charges at the solid-liquid interface create an additional driving force, pulling photogenerated electrons to the surface and driving the charge transfer reaction.
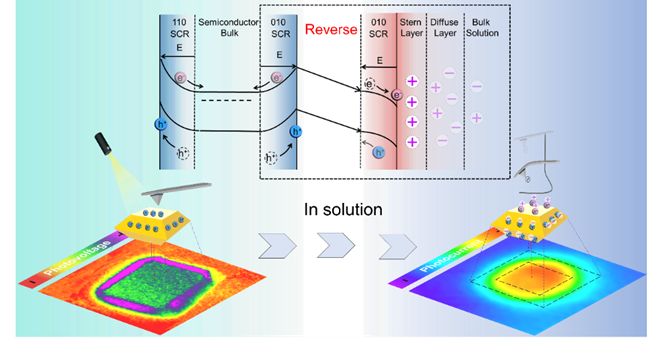
Surface charge-induced interface electric field reversal and comparative imaging in air vs. electrolyte environments (Image by LI Qian and CUI Junhao)
Researchers quantitatively demonstrated how local surface potential in the electrolyte varies with pH, providing micro-nano scale observations. By relating the surface potential and reaction product flux, they demonstrated that the photocatalytic oxygen evolution reaction rate is controlled by the surface charge induced electric field.
Additionally, researchers identified the optimal pH range for efficient spatial separation of photogenerated electrons and holes. They visualized the entire charge transfer process from the space charge region to the active reaction sites.
"This imaging framework offers a platform to directly measure surface potential and reaction current under operational conditions, providing idea into photocatalytic charge transfer kinetics at the nanoscale," said Prof. FAN. "It provides new avenues for designing efficient photocatalysts and optimizing reaction conditions," added Prof. FAN.
"Our findings provide valuable understandings into addressing the bottleneck issues of photocatalytic reactions," said LI Can.