Research group led by Prof. CHEN Ping from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences was recently invited to share their views on hydrides. This article was published on Joule.
Some important chemical transformations mediated by hydrides.(Image by WANG Qianru)
Hydrogen is the essential element having profound significance to the evolution of the solar system and the human’s understanding of the physical word. Having an intermediate Pauling electronegativity of 2.2, H (1s1) can combine with one more electron forming H ̄ (1s2).
The corresponding hydrides in the forms of molecules, clusters, surface species and bulk-phase materials are composed of one or more hydridic hydrogen atoms bonded to a more electropositive element or group.
They retain some of the characteristic features of H ̄, i.e., highly energetic, reductive, reactive etc., and exhibit unique and unprecedented chemical and physical properties that are essential to energy storage and utilization, and chemical processing including fuel production, the important fields that gain strong momentum in recent years.
Hydrides, as the hydrogen or energy carrier, have been extensively and intensively explored for hydrogen and thermal energy storage. Some hydrides were found to possess high Li+, Na+ or H ̄ ionic conductivity, allowing them to be potential solid electrolytes. More encouragingly, a few hydrogen-rich hydrides have been recently discovered as potential high-temperature superconductors.
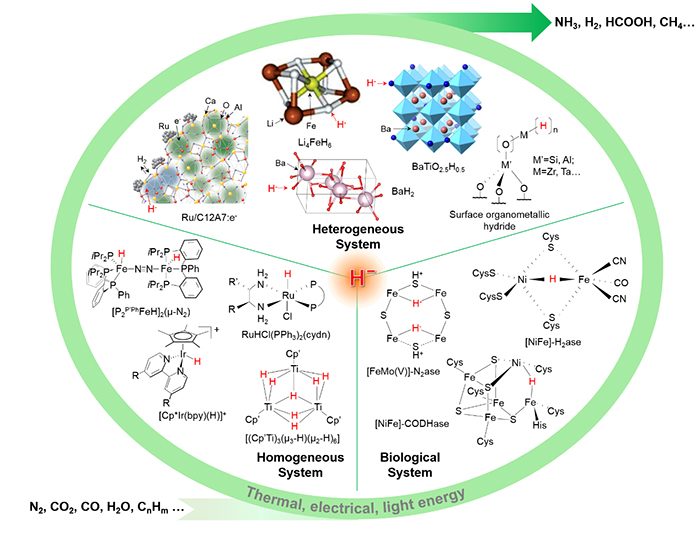
Of particular importance to our sustainable energy future is the thermal, electrochemical or photochemical synthesis of energy carriers (such as H2, CH3OH, HCOOH, CH4 and NH3) effectively from kinetically or thermodynamically stable molecules (such as H2O, CO2, and N2).
Some critical reactions that produce fuels, such as hydrogen evolution reaction (HER), N2 reduction reaction (NRR) and CO2 reduction reaction (CO2RR), generally rely on electron, proton and energy inputs.
In this context, the interchange among H ̄, H0 and H+ endows hydrides a variety of opportunities to be actively involved in such important and challenging chemical transformations.
For example, hydride, a combined source of electron and proton, plays critical roles in the transformation of dinitrogen molecule to ammonia in the thermal, biological, organometallic, and electro(photo)chemical processes.
What else can hydrides do for our energy future? It also has been shown that they offer nascent potential towards neutron shielding, light harvesting and perhaps controversial cold fusion. The story of hydrides will continue.
Future Energy articles of Joule are a forward-looking platform to highlight developing research areas which show promise in next-generation technologies and solutions, written by an expert in the field. Prof. CHEN’s group has devoted to the development of hydrides in hydrogen storage and catalytic conversion for nearly twenty years, enriching the contents of hydrides.
This work was supported by National Natural Science Foundation of China. (Text WANG Qianru)