A research group led by Prof. HAN Keli from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) revealed the new ultrafast UV energy dissipation mechanisms of plant sunscreen.
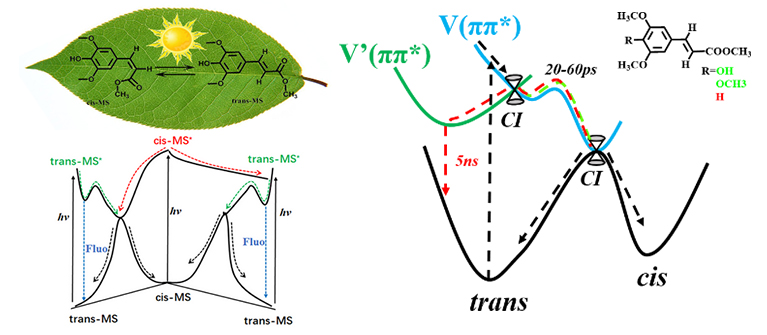
Schematic of the relaxation scheme of plant sunscreens in this work. (Image by ZHAO Xi and LUO Jian)
An overexposure to ultraviolet (UV) irradiation has deleterious effects on animals and plants. As a result, sinapoyl malate is deposited in the leaf epidermis of Arabidopsis plants and served as a sunscreen to prevent the penetration of deleterious ultraviolet irradiation. After absorption of UV light, sinapoyl malate undergoes a trans-cis nonradiative isomerization in dozens of picoseconds and results in the formation of cis product.
The scientists previously determined that the UV absorption ability of cis product was very important for the efficiency of this plant sunscreen. However, the excited-state relaxation path of the cis product remained unclear.
They used femtosecond transient absorption spectroscopy and various theoretical methods, including time-dependent functional theory, to explore the excited-state dynamics of cis-methyl sinapate (cis-MS), which was a simplified model of sinapates.
In addition to a non-adiabatic cis-trans isomerization path resulting in the formation of ground-state trans-MS, they found an adiabatic path where the excited-state cis-MS could form excited-state trans-MS directly.
This work provides a deep insight into the photoisomerization dynamics of plant sunscreen and was published in The Journal of Physical Chemistry Letters.
Furthermore, in order to construct a structure-function relationship of the sinapate sunscreens, the scientists made chemical modifications on the para-substitution of trans-MS and then explored their excited-state dynamics.
They found that after the para-hydroxyl group of MS was substituted by a hydrogen atom, a new dark excited state (denoted as V'(ππ*)) participated in the relaxation of initially populated bright V(ππ*) state. The nonradiative trans-cis photoisomerization evolved along the V(ππ*) state and was completed in about 20 picoseconds. In contrast, the V'(ππ*) state relaxed to the ground state mainly by emitting a photon and exhibited a lifetime as long as 5 nanoseconds, making it susceptible to the reactive chemicals in environment.
This is the first time to distinguish the V(ππ*) and V'(ππ*) states in experiment, giving a deep understanding of the photoisomerization mechanism of sinapates, as well as cinnamates. In addition, this work, also published in The Journal of Physical Chemistry Letters, suggests that the V'(ππ*) state should be avoided when designing a sinapate-based sunscreen.
The above work was supported by the National Key Research and Development Program of China. (Text by ZHAO Xi and LUO Jian)